Description
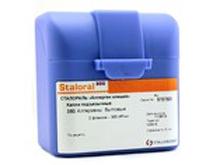
Release form
A clear, colorless to dark yellow solution.
Packaging
10 ml of allergen containing 10 TS / ml and 300 TS / ml in glass bottles with a capacity of 14 ml closed with rubber stoppers, sealed with aluminum caps with plastic caps in blue (10 TS / ml) and violet (300 TS / ml )
The kit consists of: 1 bottle with allergen 10 TS / ml, 2 bottles with allergen 300 TS / ml and three dispensers, or 2 bottles with allergen 300 TS / ml and two dispensers in a plastic box with instructions for use.
Pharmacological action of
Pollen tree allergens
IMMUNOBIOLOGICAL PROPERTIES
The exact mechanism of action of the allergen during allergen specific immunotherapy (ASIT) has not been fully studied. The following biological changes are proven:
the appearance of specific antibodies (IgG4), which play the role of blocking antibodies
a decrease in the level of specific IgE in plasma
a decrease in the reactivity of cells involved in the allergic reaction
an increase in the activity of interaction between Th2 and Th1, leading to a positive change in production cytokines (a decrease in IL-4 and an increase in -interferon) that regulate the production of IgE.
Conducting ASIT also inhibits the development of both the early and late phases of an immediate allergic reaction.
Indications
Allergen specific immunotherapy (ASIT) for patients with type 1 allergic reaction (IgE mediated), rhinitis, conjunctivitis, mild or moderate seasonal bronchial asthma, with increased sensitivity to birch pollen.
Immunotherapy can be carried out for adults and children from 5 years of age.
Contraindications
Hypersensitivity to one of the excipients (see list of excipients)
Autoimmune diseases, immunocomplex diseases, immunodeficiencies
Malignant neoplasms
Uncontrolled or severe bronchial asthma local therapy in ophthalmology)
Severe inflammatory diseases of the oral mucosa, for example, erosive-ulcerative form of lichen planus, m icoses.
Use during pregnancy and lactation
Pregnancy
You should not start ASIT during pregnancy.
If pregnancy occurs at the first stage of treatment, then therapy should be discontinued. If pregnancy arose during the period of maintenance therapy, the doctor should evaluate the possible benefits of ASIT based on the general condition of the patient.
No adverse effects reported with ASIT in pregnant women.
Breastfeeding
It is not recommended to start the course of ASIT during breastfeeding.
If a woman continues to undergo ASIT during lactation, no unwanted symptoms or reactions are expected in children.
There is no clinical data on the use of the drug during lactation.
Ingredients
Active ingredient: birch pollen allergen extract 10 TS / ml *, 300 TS / ml
Sodium chloride
rd glycerol,
mannitol,
purified water
* TS / ml – Reactivity Index – a biological standardization unit.
Dosage and administration
ASIT is more effective when treatment is started early in the course of the disease.
Dosage and regimen
The dosage and regimen of the drug are the same for all ages, but can be changed depending on the individual reactivity of the patient.
The attending physician adjusts the dosage and treatment regimen in accordance with the possible symptomatic changes in the patient and the individual reaction to the drug.
It is advisable to start treatment no later than 2-3 months before the expected flowering season and continue throughout the flowering period.
Treatment consists of two stages: initial and maintenance therapy.
1. Initial therapy is started with daily administration of a drug with a concentration of 10 TS / ml (blue bottle cap) with a single tap on the dispenser and gradually increases the daily dosage to 10 clicks. One click on the dispenser is about 0.1 ml of the drug.
Then we proceed to daily administration of the drug at a concentration of 300 TS / ml (purple cap of the bottle), starting with one press and gradually increasing the number of clicks to the optimum (well tolerated by the patient). The first stage can last 9-21 days. During this period, the maximum dosage is achieved, individual for each patient (from 4 to 8 taps daily of a preparation of a concentration of 300 TS / ml), after which they proceed to the second stage.
2. Maintenance with a constant dose using a vial of concentration 300 RI / ml.
The optimal dose achieved at the first stage of initial therapy is continued to be taken at the second stage of maintenance therapy.
Recommended dosing schedule: 4 to 8 taps on the dispenser daily or 8 taps 3 times a week.
Duration of treatment
Allergen specific immunotherapy is recommended for the above two-stage courses (2-3 months before the expected flowering season before the end of the season) for 3-5 years.
If after treatment no improvement has occurred during the first flowering season, the feasibility of ASIT should be reviewed.
How to use
Before taking the drug, make sure that:
has not expired.
is using a vial of the correct concentration.
The drug is recommended to be taken in the morning before breakfast.
The drug should be dripped directly under the tongue and held for 2 minutes, then swallow.
Children are advised to use the drug with the help of adults.
To ensure the safety and preservation of the drug, the bottles are hermetically sealed with plastic caps and sealed with aluminum caps.
Interruption in
medication If you skip taking the medication for a long time, you should consult your doctor.
If the pass in taking the drug was less than one week, it is recommended to continue treatment without changes.
If the skipping in taking the drug was more than one week at the initial stage or with maintenance therapy, it is recommended that the treatment be performed again with one click on the dispenser, using the same concentration of the drug (as before the break), and then increase the number of clicks according to the scheme of the initial stage therapy to an optimal well-tolerated dose.
Side effects
Conducting ASIT can cause adverse reactions, both local and general.
Dosage and treatment regimen may be reviewed by the attending physician in case of an individual reaction or changes in the general condition of the patient.
Local reactions:
oral: itching in the mouth, swelling, discomfort in the mouth and throat, disruption of the salivary glands (increased salivation or dry mouth)
gastroenterological reactions: abdominal pain, nausea, diarrhea.
Usually these symptoms quickly disappear, and there is no need to change the dosage and treatment regimen. In case of frequent occurrence of symptoms, the possibility of continuing therapy should be reviewed.
General reactions are rare:
rhinitis, conjunctivitis, asthma, urticaria require symptomatic treatment with H1 antagonists, beta-2 mimetics, or corticosteroids (oral). The doctor should review the dosage and treatment regimen or the possibility of continuing ASIT.
in extremely rare cases, generalized urticaria, angioedema, laryngeal edema, severe asthma, anaphylactic shock are possible, which requires the cancellation of ASIT.
Rare side effects not related to Ig-E mediator reactions:
asthenia, headache
exacerbation of preclinical atopic eczema
delayed reactions like serum sickness with arthralgia, myalgia, urticaria, nausea, adenopathy, which requires
All side effects should be reported to your healthcare provider.
Drug Interaction
Do not use concurrently with beta blockers.
Simultaneous intake with symptomatic anti-allergic drugs (H1-antihistamines, beta-2 mimetics, corticoids, mast cell degranulation inhibitors) is possible for better tolerability of ASIT.
Overdose
In the event of exceeding the prescribed dose, the risk of side effects increases, requiring symptomatic treatment.
Storage conditions
Store and transport at a temperature of 2 to 8 ° C.
Keep out of the reach of children.
The Expiration of
is 36 months. Do not use after the expiry date.
Deystvuyuschee substances
Allergen p lts berez
Dosage form
Dosage form
drops for oral administration
Stallerzhen, France