Description
Latin name
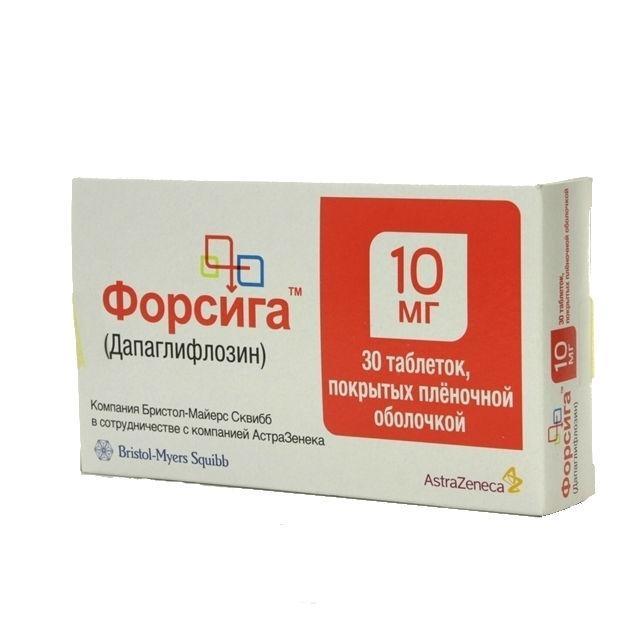
FORXIGA
Release form
Yellow film-coated tablets, diamond-shaped, biconvex, with an engraving of 10 on one side and 1428 on the other side.
Pharmacological action
Oral hypoglycemic drug.
Mechanism of Action
Dapagliflozin is a potent (inhibitory constant (Ki) 0.55 nM), a selective reversible type 2 sodium glucose cotransporter inhibitor (SGLT2). SGLT2 is selectively expressed in the kidneys and is not found in more than 70 other body tissues (including in the liver, skeletal muscle, adipose tissue, mammary glands, bladder and brain). SGLT2 is the main carrier involved in glucose reabsorption in the renal tubules.
Glucose reabsorption in the renal tubules in patients with type 2 diabetes mellitus (T2DM) continues despite hyperglycemia. By inhibiting the renal transfer of glucose, dapagliflozin reduces its reabsorption in the renal tubules, which leads to the excretion of glucose by the kidneys. The result of dapagliflozin is a decrease in fasting glucose and after eating, as well as a decrease in the concentration of glycosylated hemoglobin in patients with type 2 diabetes.
Glucose excretion (glucosuric effect) is observed after taking the first dose of the drug, persists for the next 24 hours, and continues throughout therapy. Glucose excreted by the kidneys due to this mechanism, depends on the concentration of glucose in the blood and on the glomerular filtration rate (GFR).
Dapagliflozin does not interfere with the normal production of endogenous glucose in response to hypoglycemia. The effect of dapagliflozin is independent of insulin secretion and insulin sensitivity. In clinical studies of Forsig ¢, an improvement in -Cell function was noted (HOMA test, homeostasis model assessment).
Kidney glucose excretion caused by dapagliflozin is associated with calorie loss and weight loss. Dapagliflozin inhibition of sodium glucose cotransport is accompanied by weak diuretic and transient natriuretic effects.
Dapagliflozin does not affect other glucose transporters that transport glucose to peripheral tissues, and exhibits more than 1,400 times greater selectivity for SGLT2 than for SGLT1, the main intestinal transporter responsible for glucose absorption.
Pharmacodynamics
After taking dapagliflozin by healthy volunteers and patients with type 2 diabetes, there was an increase in the amount of glucose excreted by the kidneys. When dapagliflozin was taken at a dose of 10 mg / day for 12 weeks, in patients with type 2 diabetes, approximately 70 g of glucose / day was excreted by the kidneys (which corresponds to 280 kcal / day). In patients with type 2 diabetes who took dapagliflozin at a dose of 10 mg / day for a long time (up to 2 years), glucose excretion was maintained throughout the course of therapy.
Kidney glucose excretion with dapagliflozin also leads to osmotic diuresis and an increase in urine volume. An increase in urine volume in patients with type 2 diabetes who took dapagliflozin at a dose of 10 mg / day, persisted for 12 weeks and was approximately 375 ml / day. The increase in urine volume was accompanied by a small and transient increase in the excretion of sodium by the kidneys, which did not lead to a change in the concentration of sodium in the blood serum.
The planned analysis of the results of 13 placebo-controlled studies showed a decrease in systolic blood pressure (SBP) of 3.7 mm Hg. and diastolic blood pressure (DBP) at 1.8 mm Hg at the 24th week of dapagliflozin therapy at a dose of 10 mg / day compared with a decrease in SBP and DBP by 0.5 mm Hg. in the placebo group. A similar decrease in blood pressure was observed during 104 weeks of treatment.
When using dapagliflozin at a dose of 10 mg / day in patients with type 2 diabetes with inadequate glycemic control and hypertension, receiving angiotensin II receptor blockers, ACE inhibitors, including in combination with another antihypertensive drug, a decrease in glycosylated hemoglobin by 3.1% and a decrease in SBP by 4.3 mm Hg were noted. after 12 weeks of therapy compared with placebo.
Indications
Type 2 diabetes mellitus in addition to diet and exercise to improve glycemic control in quality:
– monotherapy
– additions to therapy with metformin, sulfonylurea derivatives (including in combination with metformin), thiazolidinediones, dipeptidyl peptidase 4 (DPP-4) inhibitors (including in combination with metformin), insulin preparations (including in combination with one or two hypoglycemic drugs for oral administration) in the absence of adequate glycemic control for this
therapy – starting combination therapy with metformin, if this therapy is advisable.
Contraindications
– type 1 diabetes mellitus
– diabetic ketoacidosis
– moderate and severe renal failure (GFR <60 ml / min / 1.73 m2) or end-stage renal failure – hereditary intolerance lactase deficiency and glucose-galactose intolerance – pregnancy – breastfeeding – children and adolescents under 18 years of age (safety and effectiveness have not been studied) – patients taking loop diuretics, or with reduced BCC, for example, due to acute diseases ( such as gastrointestinal diseases) – elderly patients aged 75 years and older (to start therapy) – increased individual sensitivity to any component of the drug. Precautions: severe liver failure, urinary tract infection, risk of decreasing BCC, elderly patients, chronic heart failure, elevated hematocrit. Use during pregnancy and lactation Due to the fact that the use of dapagliflozin during pregnancy has not been studied, the drug is contraindicated in this category of patients. If pregnancy is diagnosed, dapagliflozin therapy should be discontinued. It is not known whether dapagliflozin and / or its inactive metabolites pass into breast milk. The risk to newborns / infants cannot be ruled out. Dapagliflozin is contraindicated in the period of breastfeeding. Special instructions Use in patients with impaired renal function The effectiveness of dapagliflozin depends on renal function, and this effectiveness is reduced in patients with moderate renal failure and is probably absent in patients with severe renal impairment. Among patients with moderate renal failure (CC <60 ml / min or estimated GFR <60 ml / min / 1.73 m2), a greater proportion of patients receiving dapagliflozin showed an increase in the concentration of creatinine, phosphorus, PTH and arterial hypotension than in patients receiving placebo. Forsig ² Ñ is contraindicated in patients with moderate or severe renal failure (CC <60 ml / min or estimated GFR <60 ml / min / 1.73 m2). Forsig ² Ñ was not studied in severe renal failure (CC <30 ml / min or estimated GFR <30 ml / min / 1.73 m2) or end-stage renal failure. It is recommended that you monitor kidney function as follows: – before starting dapagliflozin therapy and at least 1 time per year subsequently – before taking concomitant medications that can reduce kidney function, and periodically subsequently – in case of impaired renal function close to moderate, at least 2- 4 times a year. If the renal function decreases below the CC value <60 ml / min or the estimated GFR <60 ml / min / 1.73 m2, dapagliflozin should be discontinued. Use in patients with impaired liver function Clinical studies have limited data on the use of the drug in patients with impaired liver function. Dapagliflozin exposure is increased in patients with severe liver dysfunction. Use in patients at risk of decreasing BCC for the development of arterial hypotension and / or electrolyte imbalance In accordance with the mechanism of action, dapagliflozin enhances diuresis, accompanied by a slight decrease in blood pressure. The diuretic effect may be more pronounced in patients with a very high concentration of glucose in the blood. Dapagliflozin is contraindicated in patients taking loop diuretics or in patients with reduced BCC, for example, due to acute diseases (such as gastrointestinal diseases). Caution should be exercised in patients for whom a decrease in blood pressure caused by dapagliflozin may be a risk, for example, in patients with a history of cardiovascular disease, in patients with a history of hypotension, receiving antihypertensive therapy, or in elderly patients. When taking dapagliflozin, careful monitoring of the BCC state and electrolyte concentration (e.g. physical examination, blood pressure measurement, laboratory tests, including hematocrit) is recommended against the background of concomitant conditions that can lead to a decrease in bcc. With a decrease in BCC, a temporary cessation of dapagliflozin is recommended until this condition is corrected. Ketoacidosis During post-marketing use of the drug, ketoacidosis was reported, incl. diabetic ketoacidosis, in patients with type 1 and type 2 diabetes mellitus, taking Forsig ² Ñ and other SGLT2 inhibitors, although a causal relationship has not been established. Forsig ² Ñ is not indicated for the treatment of patients with type 1 diabetes. Forsiga ² Ñ patients with signs and symptoms indications of ketoacidosis, including nausea, vomiting, abdominal pain, malaise, and shortness of breath, should be checked for ketoacidosis, even if the blood glucose concentration is below 14 mmol / L. If ketoacidosis is suspected, consideration should be given to discontinuing or temporarily discontinuing the use of the drug Forsiga ² Ñ and immediately examine the patient. Factors predisposing to the development of ketoacidosis include low functional activity of -cells due to impaired pancreatic function (e.g. type 1 diabetes, pancreatitis or a history of pancreatic surgery), reduced insulin dose, reduced caloric intake or increased need insulin due to infections, diseases or surgery, as well as alcohol abuse. Forsig ® should be used with caution in such patients. Urinary Tract Infections When analyzing pooled data on dapagliflozin use, up to 24 weeks of urinary tract infection were more likely to occur with dapagliflozin 10 mg compared with placebo. The development of pyelonephritis was noted infrequently, with a similar frequency in the control group. Kidney glucose excretion may be associated with an increased risk of urinary tract infections, therefore, in the treatment of pyelonephritis or urosepsis, the possibility of temporary discontinuation of dapagliflozin therapy should be considered. Urosepsis and pyelonephritis. In post-marketing use of the drug, serious urinary tract infections, including urosepsis and pyelonephritis, requiring hospitalization of patients, have been reported, taking Forsig ² Ñ and other SGLT2 inhibitors. Therapy with SGLT2 inhibitors increases the risk of developing urinary tract infections. Patients should be monitored for signs and symptoms of urinary tract infections and, if indicated, should be treated immediately. Elderly patients Elderly patients are more likely to have impaired renal function and / or the use of antihypertensive drugs that can affect renal function, such as ACE inhibitors and type 1 angiotensin receptor antagonists (ARA). For elderly patients, the same recommendations for renal dysfunction apply as for all patient populations. In a group of patients 65 years of age, a larger proportion of patients receiving dapagliflozin developed adverse reactions, associated with impaired renal function or renal failure compared with placebo. The most common adverse reaction associated with impaired renal function was an increase in serum creatinine concentration, most cases were transient and reversible. In elderly patients, the risk of a decrease in BCC may be higher, and diuretics are more likely to be taken. In a larger proportion of patients 65 years of age who received dapagliflozin, adverse reactions were associated with a decrease in bcc. Experience with the use of the drug in patients aged 75 years and older is limited. It is contraindicated to start dapagliflozin therapy in this population. Chronic heart failure Experience with the use of the drug in patients with chronic heart failure I-II FC according to NYHA classification is limited, and in clinical trials, dapagliflozin was not used in patients with NYHA FC III-IV chronic heart failure. Increased hematocrit When using dapagliflozin, an increase in hematocrit was observed, and therefore caution should be exercised in patients with increased hematocrit. Assessment of urinalysis Due to the mechanism of action of the drug, the results of urine analysis for glucose in patients taking Forsig ² Ñ will be positive. Impact on the determination of 1,5-anhydroglucitol Evaluation of glycemic control using the determination of 1,5-anhydroglucitol is not recommended, as measuring 1,5-anhydroglucitol is an unreliable method for patients taking SGLT2 inhibitors. Alternative methods should be used to evaluate glycemic control. Influence on the ability to drive vehicles and control mechanisms No studies have been conducted on the effects of dapagliflozin on the ability to drive vehicles and mechanisms. Composition 1 tab. dapagliflozin propanediol monohydrate 12.3 mg, which corresponds to the content of dapagliflozin 10 mg Excipients: microcrystalline cellulose – 171.45 mg, anhydrous lactose – 50 mg, crospovidone – 10 mg 3, srdl75 magnesium stearate – 2.5 mg. Shell composition: Opadray® II yellow – 10 mg (polyvinyl alcohol partially hydrolyzed – 4 mg, titanium dioxide – 2.35 mg, macrogol 3350 – 2.02 mg, talc – 1.48 mg, yellow iron oxide dye – 0.15 mg). Dosage and administration The drug is taken orally, regardless of food intake. Monotherapy: the recommended dose of Forsig ¢ is 10 mg 1 time / day. Combination Therapy: the recommended dose of Forsig ¢ is 10 mg 1 time / day in combination with metformin, sulfonylurea derivatives (including in combination with metformin), thiazolidinediones, DPP-4 inhibitors (including in combination with metformin), drugs insulin (including in combination with one or two hypoglycemic drugs for oral administration). In order to reduce the risk of hypoglycemia with the joint use of the drug Forsig ¢ with insulin preparations or drugs that increase the secretion of insulin (for example, with a sulfonylurea derivative), it may be necessary to reduce the dose of insulin drugs or drugs that increase the secretion of insulin. Starting combination therapy with metformin: the recommended dose of Forsig ¢ is 10 mg 1 time / day, the dose of metformin is 500 mg 1 time / day. In case of inadequate glycemic control, the dose of metformin should be increased. In case of impaired liver function of mild or moderate severity, there is no need to adjust the dose of the drug. For patients with severe liver dysfunction, an initial dose of 5 mg is recommended. With good tolerance, the dose can be increased to 10 mg. The effectiveness of dapagliflozin depends on renal function. In patients with impaired renal function of moderate severity, the effectiveness of treatment is reduced, and in patients with severe impairment, it is most likely absent. Forsig ¢ is contraindicated in patients with moderate to severe renal failure (CC <60 ml / min or GFR <60 ml / min / 1.73 m2) or with end-stage renal failure. In case of impaired renal function, dose adjustment is not required. Safety and effectiveness of dapagliflozin in children and adolescents under the age of 18 have not been studied. In elderly patients, dose adjustment is not required. However, when choosing a dose, it should be borne in mind that renal dysfunction and the risk of a decrease in bcc are more likely in this category of patients. Since the clinical experience of using the drug in patients aged 75 years and older is limited, it is contraindicated to start dapagliflozin therapy in this age group. Side effects of A pre-planned analysis of the pooled data included the results of 12 placebo-controlled studies in which 1193 patients took 10 mg dapagliflozin and 1393 patients received placebo. The overall incidence of adverse events (short-term therapy) in patients taking 10 mg dapagliflozin was similar to that in the placebo group. The number of adverse events leading to discontinuation of therapy was small and balanced between treatment groups. The most common adverse events leading to the discontinuation of dapagliflozin therapy at a dose of 10 mg were an increase in blood creatinine concentration (0.4%), urinary tract infections (0.3%), nausea (0.2%), dizziness (0.2%) and rash (0.2%) ) One patient taking dapagliflozin showed the development of an adverse event from the liver with a diagnosis of drug hepatitis and / or autoimmune hepatitis. The most common adverse reaction was hypoglycemia, the development of which depended on the type of underlying therapy used in each study. The incidence of episodes of mild hypoglycemia was similar in treatment groups, including placebo. Description of individual adverse reactions Hypoglycemia The incidence of hypoglycemia depended on the type of underlying therapy used in each study. In studies of dapagliflozin as monotherapy, combination therapy with metformin for up to 102 weeks, the incidence of mild hypoglycemia was similar (<5%) in treatment groups, including placebo. In all studies, episodes of severe hypoglycemia were infrequent, and their frequency was comparable between the dapagliflozin and placebo groups. Decrease in bcc Adverse reactions associated with a decrease in bcc (including reports of dehydration, hypovolemia or arterial hypotension) were observed in 0.8% and 0.4% of patients taking dapagliflozin 10 mg and placebo, respectively, serious reactions were observed in <0.2% of patients and they were comparable in the dapagliflozin 10 mg and placebo groups. Vulvovaginitis, balanitis and similar genital infections Vulvovaginitis, balanitis and similar genital infections were observed in 4.8% and 0.9% of patients taking dapagliflozin 10 mg and placebo, respectively. Most infections were mild or moderate, the initial course of standard therapy was effective, and therefore patients rarely stopped taking dapagliflozin. These infections more often developed in women (6.9% and 1.5% with dapagliflozin and placebo, respectively), and in patients with a history of such infections they recur more often. Urinary Tract Infections Urinary Tract Infections were more common with dapagliflozin 10 mg than with placebo (4.3% versus 3.7%, respectively). Most infections were mild or moderate, the initial course of standard therapy was effective, and therefore patients rarely stopped using dapagliflosin. These infections more often developed in women, and in patients with a history of such infections they recur more often. Parathyroid hormone (PTH) A slight increase in serum PTH concentration was noted, and to a greater extent in patients with higher initial PTH concentrations. Studies of bone mineral density in patients with normal renal function or mild renal impairment did not reveal bone loss during one year of therapy. Malignant tumors In clinical studies, the overall proportion of patients with malignant or unspecified tumors was similar in the dapagliflozin group (1. 47%) and the placebo / comparison group (1.35%). According to animal studies, the drug did not show carcinogenic or mutagenic properties. When considering the development of tumors of various organ systems, the relative risk associated with dapagliflozin was higher than 1 for some tumors (bladder, prostate, mammary gland) and lower than 1 for others (for example, blood and lymphatic system, ovaries, urinary system) generally without an increased risk of developing tumors associated with dapagliflozin. The increased / decreased risk was not statistically significant for any organ system. Given the lack of information about the development of tumors in preclinical studies, as well as the short latent period between the first exposure of the drug and the diagnosis of the tumor, a causal relationship is assessed as unlikely. Because the numerical imbalance of tumors of the breast, bladder and prostate gland requires special attention, the study of this issue will be continued in the framework of post-registration studies. Elderly patients (? 65 years old) Adverse reactions associated with impaired renal function or renal failure have been reported in 2.5% of patients receiving dapagliflozin and in 1.1% of patients receiving placebo in the group of patients? 65 years of age. The most common adverse reaction associated with impaired renal function was an increase in serum creatinine concentration. Most of these reactions were transient and reversible. Among patients? 65 years of age, a decrease in BCC, most often recorded as arterial hypotension, was noted in 1.5% and 0. 4% of patients taking dapagliflozin and placebo, respectively. Drug Interactions Pharmacodynamic Interactions Dapagliflozin may enhance the diuretic effect of thiazide and loop diuretics and increase the risk of dehydration and hypotension. Hypoglycemia may occur with insulin and drugs that increase insulin secretion. Therefore, in order to reduce the risk of hypoglycemia with the joint administration of the drug Forsig ¢ with an insulin preparation or a drug that increases the secretion of insulin, it may be necessary to reduce the dose of the insulin drug or a drug that increases the secretion of insulin. Pharmacokinetic interaction Metabolism of dapagliflozin is mainly carried out by glucuronide conjugation under the influence of UGT1A9. During in vitro studies, dapagliflozin did not inhibit the isoenzymes of the cytochrome P450 system CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP3A4, and did not induce CYP2A6, CYP2A4, or isoenzymes. In this regard, the effect of dapagliflozin on the metabolic clearance of concomitant drugs that are metabolized by these isoenzymes is not expected. Effect of other drugs on dapagliflozin Interaction studies involving healthy volunteers, mainly those who took a single dose of the drug, showed that metformin, pioglitazone, sitagliptin, glimepiride, voglibose, hydrochlorothiazide, bumetanide, valsartinapinfinvine do not have any effects. After the combined use of dapagliflozin and rifampicin, inducer of various active transporters and enzymes that metabolize drugs, a 22% decrease in systemic exposure (AUC) of dapagliflozin was noted in the absence of a clinically significant effect on the daily excretion of glucose by the kidneys. It is not recommended to adjust the dose of the drug. A clinically significant effect when used with other inducers (e.g. carbamazepine, phenytoin, phenobarbital) is not expected. After the combined use of dapagliflozin and mefenamic acid (UGT1A9 inhibitor), a 55% increase in systemic exposure of dapagliflozin was noted, but without a clinically significant effect on the daily excretion of glucose by the kidneys. It is not recommended to adjust the dose of the drug. The effect of dapagliflozin on other drugs In studies of interactions involving healthy volunteers, mainly once dosed, dapagliflozin did not affect the pharmacokinetics of metformin, pioglitazone, sitagliptin, glimepiride, hydrochlorothiazide, bumetanide, valsartan, digoxin (P-gp substrate) or warfarin (S-warfarin, CYL-2, MHO. The use of dapagliflozin in a single dose of 20 mg and simvastatin (a substrate of the isoenzyme CYP3A4) led to an increase in the AUC of simvastatin and a 31% AUC of simvastatin acid. Increased exposure to simvastatin and simvastatin acid is not considered clinically significant. Other interactions The effects of smoking, diet, herbal medication, and alcohol use on the pharmacokinetics of dapagliflozin have not been studied. Overdose Dapagliflozin is safe and well tolerated by healthy volunteers at single doses up to 500 mg (50 times the recommended dose). Glucose was determined in the urine after administration of the drug (at least within 5 days after the dose of 500 mg), with no cases of dehydration, arterial hypotension, electrolyte imbalance, clinically significant effect on the QTc interval. The incidence of hypoglycemia was similar to that seen with placebo. In clinical studies in healthy volunteers and patients with T2DM, who took the drug once in doses up to 100 mg (10 times the maximum recommended dose) for 2 weeks, the incidence of hypoglycemia was slightly higher than when taking placebo and was independent of placebo. doses. Frequency of development of undesirable phenomena, including dehydration or arterial hypotension, was similar to the frequency in the placebo group, with no clinically relevant, dose-dependent changes in laboratory parameters, including serum electrolyte and biomarker renal function. Treatment: In case of overdose, supportive therapy should be performed taking into account the patient’s condition. The elimination of dapagliflozin by hemodialysis has not been studied. Storage Conditions The product should be stored out of the reach of children at a temperature not exceeding 30 ° C. Shelf life 3 years Deystvuyushtee substance Dapagliflozin Prescription drugstore prescription dosage form tablets Possible product names Forsyga tablets coated. 10 mg 30 pcs. AstraZeneca, UK